- About us
- Research
- Students & Teaching
- Seminars & Events
- Directories
- Booking Rooms & Equipment
- עברית
Home » Evolutionary study of cnidocytes (‘stinging cells’) - Dr. Yehu Moran
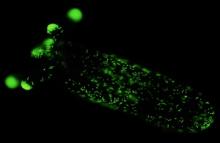
Cnidocytes (‘stinging cells’) are specialized cells that define the phylum Cnidaria (sea anemones, jellyfish, corals and hydras). They contain an “explosive” organelle called cnidocyst that acts as a 600 million-years-old microscopic injection system and is important for prey capture and anti-predator defense. These cnidocysts are the ones that used by jellyfish when it stings humans.
The arsenals of venom and the venom producing-cells are highly complex: even the structure of the delivery system- the nematocyst, is considered one of the most complex intracellular structures in nature. Our main purpose was to understand how this complexity is encoded at the genetic level.
We used the innovative CRISPR/Cas9 system to generate a transgenic line of the sea anemone Nematostella vectensis, that expresses a fluorescent protein in its cnidocytes. The transgenic line enabled us isolating and characterizing the developing cnidocytes. We discovered that cnidocyte formation is a highly dynamic and complex process in which there are dramatic shifts in the transcriptional and translational profiles of the maturating cnidocytes.
This work identified numerous novel cnidocyte-specific genes that exhibited spatiotemporal variation in their expression which indicate there are still uncharacterized populations of cnidocytes. We also revealed and characterized Cnido-Jun and Cnido-Fos1, novel cnidocyte specific transcription factors that originated via duplication and recruitment of proteins from a bilaterian stress response complex that play a significant role in cnidocyst formation.
This work reveals the evolutionary ingenuity and novelty associated with the formation of the morphologically and biochemically complex cnidocytes and provides novel insights into the biology, development and evolution of this cell-type as it highlights the recruitment not only of genes originally involved different processes such as stress response to the cnidocyte but also of novel cnidocyte-specific genes.