- About us
- Research
- Students & Teaching
- Seminars & Events
- Directories
- Booking Rooms & Equipment
- עברית
Home » Dr. Uri Gat - from Nematostella to cancer research
Collagen triple helix repeat containing protein 1 (Cthrc1) is a secreted glycoprotein reported to regulate collagen deposition and to be linked to the Transforming growth factor β/Bone morphogenetic protein and the Wnt/planar cell polarity pathways. It was first identified as being induced upon injury to rat arteries and was found to be highly expressed in multiple human cancer types.
We explored the phylogenetic and evolutionary trends of this metazoan gene family, previously studied only in vertebrates. We identified Cthrc1 orthologs in two distant cnidarian species, the sea anemone Nematostella vectensis and the hydrozoan Clytia hemisphaerica, both of which harbor multiple copies of this gene.
We found that Cthrc1 clade-specific diversification occurred multiple times in cnidarians as well as in most metazoan clades where we detected this gene. Many other groups, such as arthropods and nematodes, have entirely lost this gene family. Most vertebrates display a single highly conserved gene, and we showed that the sequence evolutionary rate of Cthrc1 drastically decreased within the gnathostome lineage. Interestingly, this reduction coincided with the origin of its conserved upstream neighboring gene, Frizzled 6 (FZD6), which in mice has been shown to functionally interact with Cthrc1.
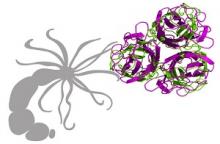
Structural modeling methods further revealed that the yet uncharacterized C-terminal domain of Cthrc1 is similar in structure to the globular C1q superfamily domain, also found in the C-termini of collagens VIII and X.
Our studies show that the Cthrc1 genes are a collagen-like family with a variable short collagen triple helix domain and a highly conserved C-terminal domain structure resembling the C1q family.